 I caught this fish in Loyalsock in 2015 and learned this week that it's probably from a hatchery. Interesting, considering the stream isn't stocked. I caught this fish in Loyalsock in 2015 and learned this week that it's probably from a hatchery. Interesting, considering the stream isn't stocked. Ask and you shall receive! My recent posts have focused on the potential influence hatchery fish can have on wild populations, particularly if they starting reproducing with one another. With stocking a common, but often contentious, practice, I knew many people would be interested in our pending data analysis and results. But, I never thought it would get as much interest as it has. Even better, I’m getting some really great questions from many of you trying to wrap your head around the pros and cons of hatchery stocking. Unfortunately, the manuscript I am in the process of writing will do very little to help clarify those questions. Heterozygosity, allelic richness, Hardy-Weinberg equilibrium, FST, genetic distance, bottlenecks …do these words mean much to you? No? That’s okay- they all refer to genetic measures that scientists look at to determine how an event (like stocking) is influencing a population. We know that certain events cause some of those numbers to go down, and other events will cause those numbers to go up. Describing how those genetic statistics have changed (or not) as a result of stocking is largely what I’ll be talking about in my manuscript. Not very reader friendly. But, those statistics are the nitty gritty. Just like you don’t need to know how an engine works to drive a car, you don’t really need to know the exact genetic details to understand the pros and cons of stocking. But, understanding some basic genetics concepts and a little lingo will go a long way in helping tease apart why the potential effects of stocking on native populations aren’t so cut and dry. It’s also helpful to have this background knowledge when deciding whether you feel the risks of stocking are worth the rewards. So, for the next three weeks I’m going to flesh out the possible pros (this week) and cons (next week), and then round out the mini-series by summarizing the consensus among biologists as to how much stocking is affecting fish populations worldwide. Where to start. Let’s first talk a little about why we have to think about genetics when assessing the effects of stocking. The brook trout of today are the product of millions of years of natural selection- genes that produce healthier fish and the most offspring are more likely to get passed on to the next generation, whereas genes that are associated with lower survival and reproduction eventually get removed from the population. Making things more complicated, the best genes for one population are unlikely to be the best genes for another population. This is called local adaptation- millions of years of natural selection have left fish from a given stream with genes that give them the best chance of survival in that stream (highlighted for extra emphasis). Local adaptation makes fish successful at living in one environment, but potentially not very successful if they are transferred into another environment. 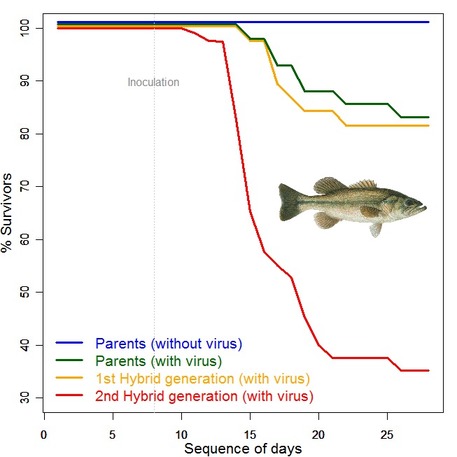 An example of the dangers of outbreeding depression- when individuals from different streams reproduce and the offspring have lower survival. Here, the yellow and red lines represent survival of the first- and second-generation offspring between two fish from different streams. You can see survival is dramatically lower than the original parents (blue and green lines). Image from conservationbytes.com. An example of the dangers of outbreeding depression- when individuals from different streams reproduce and the offspring have lower survival. Here, the yellow and red lines represent survival of the first- and second-generation offspring between two fish from different streams. You can see survival is dramatically lower than the original parents (blue and green lines). Image from conservationbytes.com. Following that line of thought, it seems like fish should stay in their home stream to fine-tune local adaptation and increase survival. But, it’s not that simple (for starters, fish don’t choose to adapt, but that’s a story for another day). Streams are highly variable environments, and local conditions change faster than fish can adapt. So, stream fish populations need to put their eggs in more than one basket- they need some fish that have genes that are successful under certain conditions and other fish that are successful in other conditions. In other words, populations need a lot of genetic diversity. With increased genetic diversity there is increased survival and resiliency to changing conditions, but also increased potential for populations to be able to adapt to future stressors (like climate change). A lot of things influence genetic diversity, but some of the biggest contributors are population size and population connectivity. Big populations that have a lot of fish moving into and out of them tend to have high genetic diversity. Historically, this is probably how many brook trout populations existed. But, times have changed. Population sizes have declined following natural disaster, disease, habitat loss, etc. And, many populations are now isolated by waterfalls, road crossings, and thermally unsuitable habitat. When populations get too low and too isolated, genetic diversity quickly erodes as fish start inbreeding and other genes get randomly removed from the population (a process known as genetic drift). For all of these reasons, one of the biggest priorities fisheries conservation managers have is to restore genetic diversity by increasing population size and connectivity. Did you follow all that? No? That’s okay. Big picture- fish become genetically specialized to the local environment (local adaptation), which increases their survival. But, the population can’t become too specialized because it needs high genetic diversity in order to be withstand disturbance and have the potential to adapt to future conditions. Okay, so where does stocking come into play? First off, there are two basic forms of fish stocking. It’s important to keep them separated as we discuss the pros and cons of stocking because they are very different from one another and have their own benefits and drawbacks. The less common form is conservation stocking, where the goal is to increase population sizes and genetic diversity of critically threatened or endangered fish or reintroduce a species to its native habitat after restoration. In conservation stocking, the fish populations are generally not harvested, and stocking is used to prevent future population declines and extinction. Conservation stocking is very tricky business. The genetics of every individual used for reproduction are carefully considered so that the stocked population is locally adapted to the wild environment, thus giving fish the best chance of surviving and reproducing. This also helps avoid outbreeding depression - when a native, locally adapted fish spawns with stocked fish that is not locally adapted and the offspring have lower survival and reproduction. Outbreeding depression has the potential to cause rapid population declines as each generation continues to have lower and lower survival and reproduction. To avoid potential effects of outbreeding depression, fish used in conservation stocking are often brought in directly from the wild and only kept in captivity for a few generations, thus minimizing genetic differences between wild and stocked populations.  Yellowfin madtom is just one example of a species being stocked for conservation. Recently populations were released into their native range in Virginia, and the success of the project is still being monitored. Photo from knoxmercury.com. Yellowfin madtom is just one example of a species being stocked for conservation. Recently populations were released into their native range in Virginia, and the success of the project is still being monitored. Photo from knoxmercury.com. Now, contrast that with recreational stocking. Recreational stocking, especially for trout, is by far the most common. The goal here is to stock as many large fish as possible in order to increase angler satisfaction. Genetics are considered, but mostly as a means to grow bigger fish, faster. In other words, fish used for recreational stocking have been artificially selected. As opposed to natural selection (where the environment picks the best genes), in artificial selection humans are the ones deciding which genes are best. It takes many years for humans to artificially select the genes that will make a population grow large and fast. But, once that goal is achieved, wild fish are no longer brought into the hatchery because wild fish will not grow as large and as fast as their artificially selected counterparts. In fact, it’s been over 100 years since the last wild fish has been introduced into many hatcheries used for recreational stocking. Because of this, fish used in recreationally stocking are genetically incompatible with the wild environment, and have lower survival and reproduction once released. So, what are the pros of stocking?
So, yes, stocking can have positive influences on native populations. But, I’ve already hinted a few times that most stocking and hatchery practices are unable to realize some of these potential benefits. But, tune in next week for a more detailed discussion about the possible negative consequences of stocking.
1 Comment
 Science may ban me now. Science may ban me now. I wrote last week of the two types of grad student vacations, conferences and field work. But, there’s another holiday that’s even rarer (at least for me) and merits even more celebration. I’m talking about your advisor’s vacation week, otherwise known as Grad Student Independence Week. Truth be told, my advisor’s whereabouts don’t really influence my work ethic. For the time being, I’m working at my own self-defined pace (cross my fingers I can keep it that way). But, the closer we get to the beginning of the semester, the more sparse the office gets. With no one to pester during the day, why bother going in? So, I didn’t. I slept in a little later (which for me is 6am), enjoyed coffee on my patio, and had one main goal: start working on the hatchery-wild hybridization manuscript. Data analysis is still on going, but at this point I know what the results are going to say. There’s no need to wait for the final numbers to crunch to start the long process of preparing the work for publication. When I was an undergrad, I always thought that scientific publications were the works of brilliant scientists who wrote the equivalent of Shakespearian prose. I never thought I’d be smart enough to accomplish a similar feat. I actually still think that, except I’ve somehow been let into that elite crowd of published scientists seven times now. It still hard to believe I’ve reached the point in my career where I am the authority on a topic- someone out there is reading my manuscript and thinking I am the brilliant scientist. Crazy. One thing I have learned along the way is that regardless of how smart you are, how great your research is, or how well you write, all manuscripts start in the same place. With a blank Word document that just stares at you. For me, it’s probably the single most intimidating and frustrating part of the publication process. Literally anything I put down “on paper” would represent an improvement over the blank page, but I just sit there for hours- staring, erasing, and getting more frustrated. There’s all sorts of advice out there about how to be the best, most efficient writer- outline your ideas, write 30 minutes every day, discuss your paper beforehand, etc.- and I defy every single recommendation. That long, frustrating, fight with the blank page is just part of my process, and I need to work through before I can write something worth saving. And, the fight needs to be long and uninterrupted. Not a great task for tackling at the office where distractions are imminent, but a perfect job for celebrating my Grad Student Independence Week at home.  One lake in Algonquin Provincial Park. Number one habitat requirement for brook trout seems to be that it is "pretty." Photo courtesy of amusingplanet.com. One lake in Algonquin Provincial Park. Number one habitat requirement for brook trout seems to be that it is "pretty." Photo courtesy of amusingplanet.com. I actually only got one full day at home, but it was enough to win the battle and get a solid start on the manuscript. Time to save it, back it up, and not look at it for at least a few days. In the meantime, I go back to square one- read published manuscripts that I know are important for my study and that I will cite in my own publication to support why our study was needed and to add credibility to the results we found. As I’ve said before, there aren’t a lot of studies on hatchery-wild interbreeding in brook trout. But, I did find one by Andrew Harbicht and colleagues (see below for a link to the manuscript) that looked at how the probability that hatchery trout will breed with wild trout changes depending on the environment. I’m still not releasing the result of our analysis, but studies like this are important regardless of what we find. Whether we find a high degree of interbreeding or not much at all, we need to know WHY we are getting that result. And, it makes sense that environmental conditions influence how much hatchery trout breed with their wild counterparts. The study was conducted on several lakes in Algonquin Provincial Park in Ontario, Canada, of which some were never stocked with hatchery brook trout, and others had historic stocking that had been stopped 10+ years prior to their study. Immediately, you’ll notice there are some differences between their study and ours: we work on streams, and in areas that are currently being stocked with high densities of fish. Nevertheless, their results are important to keep in mind as we move forward. Most importantly, they found:
So, why is this study important for us? For starters, streams often support lower populations of brook trout than lakes, making us nervous that interbreeding may be more prevalent in streams than lakes- particularly, again, because stocking in our systems is frequent and on going. Our streams also have a wide range accessibility, pH, and other environmental variables (e.g., gradient and temperature) that influence population sizes and competition. Big picture, this study just shows us that introgression isn’t an all or nothing phenomena. Location matters a whole lot, and our results can’t be taken as the definitive response of trout to stocking. But, all of this presumes that we are finding interbreeding. Which I’m not saying we are. I’m also not saying we aren’t. You’ll just have to stay tuned. *Note: Content in this post is my own and may not reflect the opinion of the manuscripts' authors or the agencies they represent. I encourage you to read the manuscript, found here, so you can contribute to the discussion.  What a difference a few years makes. You'd never know the stream was completely leveled and grew back. And the photo on the bottom, courtesy of http://goth-gardening.blogspot.com, was from 2015! What a difference a few years makes. You'd never know the stream was completely leveled and grew back. And the photo on the bottom, courtesy of http://goth-gardening.blogspot.com, was from 2015! In grad school there are two types of “vacations.” There are conferences, which you hope are in desirable cities that you can explore at night after you’ve turned your brain to mush hearing about cool research and talking to collaborators all day. And then there’s volunteer field work, where you are probably working harder than you would had you not gone on “vacation,” but are just happy to be seeing a different system and learning about a different project for a little bit. Right now, I’m on the later form of vacation. I traveled six hours south to my alma mater in Ashland, Virginia to help put another year of Mechumps Creek post-restoration monitoring into the books. Some of you may recall Mechumps Creek from my post last year, where I described the need to rebuild this urban stream to restore and protect habitat from stormwater runoff. And, no, there are no trout in central Virginia. But, had it not been for a class on stream restoration featuring this tiny creek, I would have never pursued a career in fisheries. So, Mechumps Creek will always hold a special place in my heart and I’ll gladly “vacation” here anytime (but ask me again when vacation loses those quotation marks). But, this year’s sampling was a bit different than past. Per the usual, we picked the hottest day of the year for fieldwork. With heat indices once again soaring to 110°F, I swear the Farmer’s Almanac could start using us for their long-term forecasts. And, there was no lack of poison ivy, dense thorns, and interesting animals. As it turns out, beavers don’t much appreciate 250 volts rolling through bodies. The difference this year was that we got to do twice the work. Lucky us! Normally, our sampling is focused on one section of stream that was restored in 2010 and is now being monitored to determine the long-term response of fish populations (among other things) to restoration. Though there are still a few years left in monitoring, restoration of the first section went so well that a grant was recently received to restore the next reach located just downstream. Woohoo! But, before restoration can start, there needs to be baseline measurements of what the stream and fish community look like now so that later they can determine if restoration was successful. So, after sampling the post-restoration section, we headed downstream to complete the pre-restoration assessment of the fish community.  The post-restoration reach shortly after the construction crews left. Soon this will be the fate of the downstream reach. The post-restoration reach shortly after the construction crews left. Soon this will be the fate of the downstream reach. I was part of pre-restoration sampling of the original section seven years ago, but I’ve long forgotten what fish we caught way back then. So, it was interesting to see, back-to-back, the difference in habitat and fish diversity between pre- and post-restoration sites. Post-restoration, the stream is, on average, only a couple feet wide with maybe five pools in the entire 1,200-foot section, the deepest of which about three feet. Compare that to the pre-restoration reach, which was much wider, much siltier, and was pool after pool, with some too deep to wade through. On the surface, the shift from many deep pools to mostly small riffles with restoration may seem a little undesirable, especially for all you trout enthusiasts out there. After all, big fish need big pools. But, not all streams are created equal, and management goals are not the same for all streams. While we may want to increase pool size and depth in a coldwater trout stream, Mechumps Creek is a tiny, warmwater system. We don’t necessarily care about the size structure of fish because no one is hoping to catch a citation sunfish out of Mechumps Creek. But, in order to preserve the integrity of large creeks and rivers downstream, we do care how well the ecosystem is functioning in Mechumps Creek. And, from a fish perspective, an indication of ecosystem function is how many different species are present (species richness) and how abundant each species is (species evenness). In short, we not only want to see many different species, we also want to see that individuals of each species are equally common throughout the stream as opposed to only one or two species dominating.  An oldie but a goodie. Couldn't help but think of this exact photo as we passed this spot sampling this year. That sand bar behind us was still intact, as are many of the defining features I remember from my time spent on the creek my freshman year. Soon, those features will only memories. An oldie but a goodie. Couldn't help but think of this exact photo as we passed this spot sampling this year. That sand bar behind us was still intact, as are many of the defining features I remember from my time spent on the creek my freshman year. Soon, those features will only memories. How do you increase both richness and evenness? By increasing habitat complexity and diversity- in this case reducing the number of deep pools and increasing the number of riffles and runs to provide habitat for many species that prefer many different types of habitat. This is one of the main goals of stream restoration that is best accomplished by reshaping the existing channel and reducing future streambank erosion (how this is accomplished is feat of skilled engineering, of which I won’t get into the details here). So far, it seems past restoration efforts at Mechumps Creek have accomplished this goal. This year, while there were many species in the downstream, pre-restoration site (i.e., moderate richness), many of them were fairly rare (low evenness). Most of the fish we caught in the pre-restoration site this year were catfish, sunfish, and mudminnows, all species that we see most commonly in muddy backwaters with low oxygen(i.e,, the habitat that was most prevalent). In the post-restoration site, we found many more species (higher richness) and improved evenness. There were fewer catfish and mudminnows, and far more darters, dace, and chubs, species that all prefer habitats with more moving water and less silt. So, we can tell from the difference between the fish communities that habitat restoration has long-term improvements to species richness and ecosystem function of Mechumps Creek. Hence the reason for moving on to phase two! This year wasn’t just special because of the ability to do pre- and post-restoration comparisons. Because we’ve focused all of our attention on the post-restoration reach the last few years, I hadn’t visited the pre-restoration reach in about ten years, back when I was a college freshman hoping to pursue a career in surgery. So much has changed since then, but walking through the stream was like rewinding the clock. Trees we used for survey benchmarks still stood strong, sandbars we had group meetings on had only grown larger, and our little foot paths through the dense brush still seemed completely intact. I can remember many cold, rainy afternoons spent roaming around that stream with my comrades, Sonni and Arba, naïve to the future to come (nor knowing what I even wanted the future to hold), but having the time of my life trying to learn about stream ecology. Now, here I am, ten years later. I’m turning the corner on my Ph.D., still naïve, but still having fun learning stream ecology. Surveying Mechumps Creek this year, I’m reminded of the leap of faith I took in deciding to pursue a career in Ecology. I won’t lie, I sometimes wonder what might have happened if I didn’t ____ (fill in the blank with any of about 100 serendipitous decisions that got me where I am today) and I had pursued medicine. But, I think back to the experiences I’ve had over the last ten years, the utterly ridiculous things I still find interesting about fish, and the curiosity I still have for research, and there’s no doubt I made the right call. I’m excited for this next phase of restoration- the restoration project I pitched to Ashland Town Council ten years ago and worked on tirelessly my entire first year of college is finally being realized. But, it’s also bittersweet. The little ecosystem that taught me a love for field ecology was practically unchanged. But, in just a few months, it will all get ripped out with a few swings from the backhoe. It’s a little like renovating your childhood home. I now it needs restoration, but some good memories and a lifetime worth of professional gratitude are tied up in those ugly, eroding banks. So, I bid farewell to pre-restoration Mechumps Creek. But, the story doesn’t stop here. I’ll go back next year to visit the new and improved stream, meet the new tenants, and start the next chapter of Mechumps Creek.  My office has a nice view My office has a nice view It seems like I was just out in Loyalsock, but the calendar doesn’t lie. Two weeks had come and gone and it was time to head back out and collect more blood samples. A full day in the woods, avoiding the ever-increasing traffic around town, handling native trout in the clear, cool stream? A pity, my life. It’s been a fairly wet summer, especially compared to the severe drought of last year. Nonetheless, streams flows have really dropped in the last few weeks and are reaching summer lows. Yet, I continue to be surprised with some fairly large brook trout in some pretty crappy habitat. But, hey, no complaints here. Work smarter, not harder, right? Or, more accurately, thank you field Gods for blessing me with good fortunes. But, it’s not just stream flows that have been changing. Over the last few weeks, the trout have put on their summer figures: long, lean, and almost unhealthy looking. How could this be? Just a few months ago I posted about how fat and “football-like” the trout were. The answer is simple. They’re starving. It’s a common misconception I often hear about stream trout. In the summer, we’re constantly getting bit by bugs, we see a bunch flying around, and we may even flip rocks and see various aquatic invertebrates crawling around. Insects are everywhere, and so it would make sense that summer would be buffet season for trout.  Far from the worst I've seen, but the fish this week were noticeably more skinny than sampling events prior. Usually it's the area around the tail that starts to loss weight the fastest. Far from the worst I've seen, but the fish this week were noticeably more skinny than sampling events prior. Usually it's the area around the tail that starts to loss weight the fastest. But, not all insects are created equal. Think about the type of bugs that you see in summer and those you might see during a spring hatch, the time of year when many invertebrate species are transitioning between aquatic larvae and terrestrial adults. Spring hatches may be brief, but the number of bugs swimming and flying around is unmatched. And, many of these bugs are floating on the surface of the stream or in the middle of the water column, making them easy targets for trout. In spring, almost all of a brook trout’s diet is comprised of these aquatic prey forms. Now, think about the bugs you see in summer. Hatches are over, and most of the invertebrates that are left in streams are difficult for trout to eat because they are glued to the bottom, hiding between rocks, and, in cases like caddisflies, armored is shells. By summer, stream trout are feeding almost entirely on terrestrial insects. But, this food source doesn’t come easy. Most terrestrial insects aren’t buzzing around the water surface for too long, and it takes a lot of energy for trout to jump out of the water to capture their prey, especially when most attempts are unsuccessful. In all, these terrestrial insects are much harder to capture, and in many cases offer fewer calories than the spring hatches. Making matters worse, as stream flows recede, habitat starts collapsing down giving fish fewer places to forage, but also fewer places to hide from increasing predation pressure. Given the choice between eating and hiding, trout will usually choose hiding until they are at the extremes of starvation. The degree of summer food deprivation certainly varies by region, and even by stream. Lakes and large rivers go largely unaffected, but in smaller streams, there is often not enough food available for fish to maintain basic metabolic functions. When this happens, fish growth stops and condition factor (the ratio of length to weight) takes a sharp drop. That’s when fish get that long, lean appearance I’ve dubbed “summer trout bod.”
Long-term food deprivation can of course result in death, but can also have long-term effects for survivors. As brook trout are fall spawners, summer is the time they should be devoting calories to production of gametes (eggs and sperm). When fish are barely meeting basic metabolic demands needed for survival, they are can’t devote energy towards gamete production. As a result, they produce fewer and lower quality gametes, leading to fewer and lower quality eggs and juveniles. It’s a vicious cycle. So, when you head out to your favorite trout stream this summer, the fish might bite like crazy. But, it’s not because they are having a alive and well. It’s probably because they are in desperate need of calories and willing to take more risks and eat a larger variety of prey than they normally would. There’s some controversy in angling if it’s ethical to take advantage of fish when they are in heightened states of vulnerability. Usually this centers around discussions of “bed fishing” (targeting bass on their spawning nests) and targeting coldwater zones that attract fish during high temperatures. But, you could certainly argue that summer trout fishing starts edging closer towards the category of giving humans the unfair advantage. I’m not here to play fishing police (I was fishing just the other day, in fact), but just something to think about next time you grab your fishing pole.  This fish, and roughly 2,100 of his cousins, proudly gave their fins for our study. This fish, and roughly 2,100 of his cousins, proudly gave their fins for our study. We turned a new page this week. After two years, our population genetics dataset is finally complete. This means I have the genotypes (essentially the genetic identity) for over 2,100 fish across the Loyalsock Creek watershed and some of the most common hatcheries that stock in the area. It’s a huge step forward. I had reached a standstill with just about every project I am working on because they all needed the genetic data for some reason or another. Now I can finally start producing some much-anticipated results. So, which project do I start with? The obvious answer is with a study that is beyond the scope of any grant and not one of the original chapters of my dissertation. The original intent of collecting population genetics data was to describe the degree of connectivity among 20 tributaries in the watershed. And, I’ll still do that. In fact, I’ll be doing it for 29 tributaries because I got a little overzealous in the summer of 2015 and just kept sampling. Oops. After running some preliminary analyses on those 29 tributaries back in December and talking to some locals about stocking events, it became clear to us that we couldn’t faithfully report any final genetic results without accounting for the potential influence of stocked fish on natural population genetics. Simple enough, but adding the hatcheries into the dataset resulted in a seven-month delay in data analysis. Thankfully, the analysis goes a little faster than the lab work, and I’ve already gotten some preliminary results on the hatchery + wild dataset. It looks like the dataset was worth the wait and some interesting things are showing up. So interesting so that we’ve decided to add another study that quantifies the amount of introgression between wild and hatchery brook trout. I’ve briefly discussed introgression in a previous blog, but in short it’s a fancy term to describe the mating between wild and hatchery fish. Generally speaking, introgression is a negative consequence of stocking. Hatchery fish often lack genetic diversity, and may carry genes that are maladaptive to natural environments. This isn’t a big problem if the stocked fish aren’t spawning because all of the maladaptive genes are removed from the population before they can get carried into the next generation. But, when hatchery fish do spawn, there can be problems. You can think of reproduction as essentially taking the average between two parents. If you take the average of a genetically rich wild fish and a genetically poor hatchery fish, the result is offspring that are genetically inferior with lower survival and reproduction when compared to offspring spawned from two wild parents. This effect, known as outbreeding depression, often happens when there is a high degree of introgression between wild and hatchery fish, and can quickly lead to population collapse.
Unfortunately, while I can tell you that hatchery fish and wild fish are genetically very different from one another, the analyses are still a little too young and the topic potentially a little too controversial, to comment on the degree of introgression we are finding. But, I can tell you why, regardless of whether we see introgression or not, these results are really interesting:
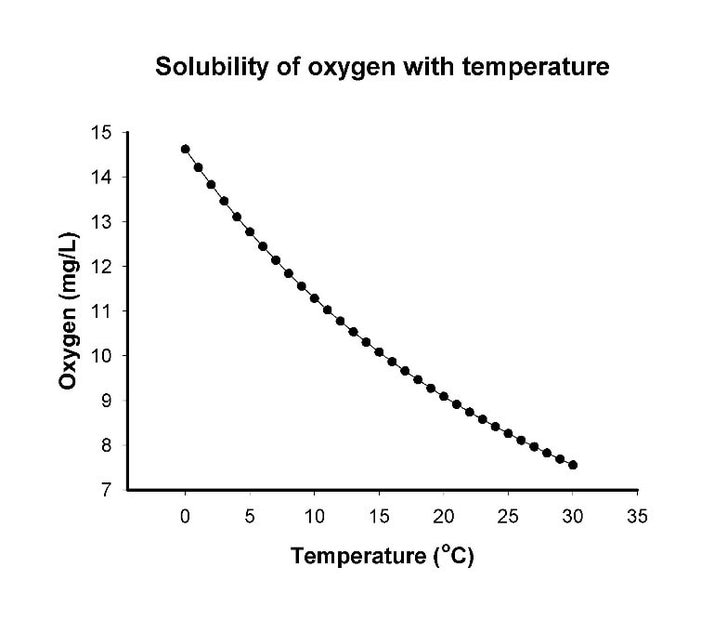
So, any predictions? Do you think Loyalsock brook trout have signs of introgression?  March sampling with a stream temperature maxing out at 32.2F. This day sucked. March sampling with a stream temperature maxing out at 32.2F. This day sucked. There’s no way around it. Sometimes my job sucks. Like days when I’m sampling in sub-freezing temperatures in November, or grabbing my gear as I hear the weather man use words like “oppressive heat warning.” There’s also 18-hour days, days spent hiking up cliffs, days where you feel like you must be wallowing in a pit of stinging nettle, poison ivy, and mosquitos, and days were all three of those events combine. And then there are days where I have the burden of electrofishing a stream loaded with 9+ inch native brook trout in the breeze of an unseasonably cool summer day. Yeah, I didn’t say my job always sucks. And, this week, my job didn’t suck. In one of the easiest field days in recent memory, we recruited 22 new brook trout into my study (yes, I know I just jinxed myself, but I’m hoping it’s retribution for some of the other days I’ve had this year). That’s 22 more blood and gill samples packed away in very deep freeze to be studied for their expression of stress proteins. In most of my field work descriptions, I’ve taken the easy way out and said I’m collecting a “blood sample.” That’s not entirely accurate. I’m collecting a blood sample, which I then use to get a plasma sample. Though it is the largest component of blood (making up about 55% of blood, by volume), plasma may seem a bit foreign to you unless you’ve donated or received it. Masked by the more obvious color of red blood cells, straw-colored plasma is practically invisible when suspended in whole blood. But, it’s made up of some of the most important elements in the body- water, salt, enzymes, and, you guessed it, proteins. This includes all the stress proteins we are interested in measuring as stream temperature rises.  The Loyalsock Fish Plasmapheresis Center. It was significantly remodeled now that we use an electrical outlet rather than the truck battery on the side of the road. The Loyalsock Fish Plasmapheresis Center. It was significantly remodeled now that we use an electrical outlet rather than the truck battery on the side of the road. With a little know-how, it’s actually quite easy to separate red blood from plasma. The folks at the American Red Cross do it with a big machine that takes your whole blood and spins it in a circle really fast (otherwise known as centrifuging). Red blood cells are heavier than plasma, so centrifuging pulls the heavier red blood cells to the bottom, and the lighter plasma components to the top. Once separated, it’s then possible to remove plasma and, in the case of the Red Cross, return red blood cell components back to the donor. In the fish world, we do things a bit differently. First, there are no big machines. There are small centrifuges running off car batteries or borrowed electricity from the Snack Shop at World’s End State Park (the view is inarguably better here than in the plasmapheresis center). And, forget return to donor, we’re greedy and keep both the plasma and red blood for analysis. But, other than that, the process is about the same. Collect blood, centrifuge blood, remove plasma, repeat. (We may also be feeding ourselves the sugary post-donation snacks. But, we checked, the fish don’t mind.) In total, it takes about three hours to remove plasma from 22 blood samples. And, what would you know, we meet a lot of people curious as to why there’s a few college kids with a centrifuge and sterile gloves next to the Snack Shop. Luckily, I like to chat fish to anyone willing to listen. And, I’ve pretty much memorized my spiel when asked “what the heck are you doing?” So, I start chatting about climate change, fish movement, and gene expression. And, you want to guess the number one question I get asked in return?  Have you found any gill lice? Seriously? I’m over here teaching a course in fish phlebotomy and toting around the equipment that allows me to electrocute fish WHILE I walk in the water, and you want to know about a parasite? Move along! In all seriousness, I do get asked some great questions about my research, and I appreciate that so many people are so attune to this rising threat. Fortunately for me, even after taking gill biopsies from over 400 fish in the last year, I have never personally encountered a trout infected with gill lice. And, I am thankful for that. What’s the big deal with gill lice? Despite the name, gill lice are not related to the form of head lice common to elementary schools and daycares. Gill lice are small crustaceans (yes, just like shrimp, crabs, and crayfish) in the genus Ergasilus. Male gill lice free-float throughout the water column and females attach to the gills of fish. That may seem innocent enough, but once attached gill lice start feeding on blood pumping through gills, thereby interfering with a fish’s ability to “breath” (absorb oxygen and release carbon dioxide). After prolonged, intense infection, fish can have reduced growth and reproduction, compromised immune systems, altered behavior, and can eventually die. Gill lice are a bit unique in that they are host-specific, meaning any given species of gill lice (of which there are several) is selective as to which species of fish it is willing to parasitize. So, there is a species of gill lice for brook trout, and a gill lice species for rainbow trout. So far, gill lice in Pennsylvania have only been shown to infect brook and rainbow trout, but other species of gill lice have been shown to infect salmon, bluegill, bass, walleye, and yellow perch, among many others.  An up-close look a few gill lice. Image courtesy of henrysfork.org. An up-close look a few gill lice. Image courtesy of henrysfork.org. While the press coverage is new, gill lice have been infecting streams throughout the United States for decades. It was only recently that gill lice started making front page headlines in Pennsylvania when a large outbreak was discovered in Centre County in 2016. The origin of the gill lice in Pennsylvania can be debated, as they could have gone undetected in wild streams for years prior to the 2016 discovery. However, there is also a smoking gun pointed towards a cooperative fish hatchery, which was known to have infected trout in the hatchery around the time of discovery. Regardless, what we know now is that gill lice have established residence in many Pennsylvania streams. And, once we started looking for gill lice we kept finding them. So, the number of streams infested with gill lice only continues to climb. How many streams are infected with gill lice? We don’t know. How are gill lice affecting trout populations in Pennsylvania? We don’t know. And, how can we stop the spread? You guessed it, we don’t know. So far, the state is trying to minimize the spread by decontaminating hatcheries and sampling streams to determine the extent of infection. It’s going to be a tough battle, though, because gill lice are resistant to chemical treatments that can be used to reduce parasite loads. So, unfortunately, managers’ hands are largely tired in trying to eradicate the parasite from wild streams. While we in Pennsylvania may be on the very start of an outbreak, we can look west to predict how trout populations may fare. Reports from Colorado and Wisconsin suggest two important things. First, trout populations decline after gill lice infestation. Second, warmer temperatures lead to higher infection rates. This could be because gill lice are more productive in warmer temperatures, trout become more susceptible at warmer temperatures, or a combination of both. What can you do to prevent the spread? As a general rule of thumb, always disinfect your gear when you’re leaving a stream. This includes hopping across major watersheds, but even if you’re just moving between neighboring tributaries. Gill lice may be present without you knowing it, and even if they aren’t you can help prevent the spread of other aquatic and terrestrial invasives. Also, it goes without saying, don’t move fish between streams, as this could result in new infections. When you’re fishing, keep an eye out for little rice-like nodules on the gills, opercula, or pectoral fins. Take a picture if you can, and report any suspicious findings to your local authorities. It’s always disheartening to put another stream on the infection list, but it will help managers contain the spread and devise a plan forward. Also, keep in mind, stream temperatures are rising and for many it’s getting close to that time of year where you should consider switching your target species. Angling mortality greatly increases at 65F and above, so take a thermometer and fish responsibly.  A coldwater seep from Benner Run near Philipsburg, Pa. A coldwater seep from Benner Run near Philipsburg, Pa. Our undergraduate research assistant, Ben Kline, continues to make impressive progress on analyzing fish behavior videos from the laboratory study we did in Leetown, West Virginia last year. This week, I’m turning the blog back over to him to share a brief introduction on why this study was done. If you missed his first post, be sure to check it out here. Looking forward to getting some results soon! It is no secret that all living organisms require a few basic resources to survive. Often when we think of these necessities we list things like food, water, oxygen, shelter, etc. While it is obvious to us that we need to meet these basic requirements to survive, we often neglect that our aquatic counterparts have needs that vary slightly from our own. My most recent project in the lab is investigating just how important one of these lesser-known resources is. There are many factors that impact the availability of essential resources in any given habitat. At a basic level, aspects of the environment, such as climate and geography, limit resources to a fairly narrow range of possible conditions. Just consider the resources available in a mountain stream and those in a coastal estuary. These systems may be fairly close to one another, but the resources are very different because of the physical location of the waterway. Then there are human practices, such as land and water use, that can further restrict resource availability and/or make habitats more hospitable to particular species. For example, dams have been known to cause considerable changes to species composition, all because of how they alter resource availability, especially flow and water temperature. Taken together, the distribution and abundance of resources have a major impact on the type and number of organisms that can inhabit a certain space. As promised in my previous post, this week’s post will detail a resource that is less understood by us, but critical to the survival of numerous trout species and other coldwater fish: thermal refuge. Thermal refuge, in its simplest form, describes the availability of a cold water in a body of water that warms. I don’t know about you, but I have taken an unintentional dip or two while fishing in late spring and early summer and I would be hesitant to say that there is any shortage of cold water available for the fish. So what’s the deal? To understand the significance of temperature in trout habitat, we should take a closer look at how water temperature shifts with the seasons. In larger bodies of water, such as lakes and large reservoirs, the issue of cold water access during summer is less pressing because of the depth of the water. In these large bodies of water, thermal stratification creates zones with varying water temperatures. At the surface, water is exposed to high summer air temperatures and heats up. And, while wind can cause it to mix and circulate, warm water stays at the surface because cold water is more dense and sinks to the bottom. Thus, the surface water warms while the deeper layers, which are largely not exposed to air, remain cool, even in the hottest parts of summer. This deep layer provides of a site of refuge for coldwater species to retreat to when the rest of the lake becomes too warm.  Plants and trees along the creek bank can provide some reprieve from the hot summer sun. Plants and trees along the creek bank can provide some reprieve from the hot summer sun. While this is great news for our lake dwellers, fish that are native to creeks and streams often find it significantly harder to find cold water during the hot summer months. As I mentioned in my last post, the shallow nature of most creeks and streams means that there is no easy way for these waters to remain cold in the summer. Being close to a cold water source, such as a spring or upwelling, can help to keep the water cool. So can the presence of a dense tree canopy or sufficient riparian vegetation. Ultimately; however, the main body of water can warm to the point that the habitat may be unfit to support trout populations for an extended period of time. Contrary to popular belief, the most serious danger in inhabiting warmer waters is not entirely due to the fact that the fish cannot tolerate warmer temperatures. All organisms have an internal set point known as a thermal maxima, which is the highest temperature an individual can tolerate before it perishes. While brook trout prefer cold water, they have actually been observed to have a thermal maxima of around 25C, which is quite warm in terms of water temperature in temperate regions of the US. In fact, it is not uncommon for trout to favor warming waters that are abundant in food supplies over cool, less productive water for a short period of time. The real danger with warm water is a little more subtle and lies in the change to the dissolved oxygen content in the water as it heats up . Trout are very sensitive to drops in oxygen, and, compared to other species of fish, do not fare well when the oxygen concentration is too low. As water heats up, the solubility of oxygen (i.e., the maximum amount of oxygen that can dissolve in water) starts declining. As you can see in the graph below, at around 20°C the solubility of oxygen is less than 55% of what it is at 5°C. In short, this means that warmer water has less oxygen. And, with less oxygen in warmer water, trout start becoming metabolically and physiologically stressed and mortality increases. But, the plot thickens even more. As mentioned above, in thermally diverse systems, there can be a trade-off between cold, oxygen-rich water and warmer waters where there is more food. Too much of either can be a bad thing, and trout have to constantly make on-going decisions about which habitat type they want to occupy at any given moment.
Often times, though, the decision may not be entirely theirs to make as cold water refuge is a very limiting resource. One of the most common sources of cold water refuge are spring-fed tributaries. These tributaries are much smaller than the main channels and provide less habitat and fewer resources to support larger populations. These zones are therefore a major source of competition among fish. There is significant evidence that shows trout populations actively uses these refuge zones during hot times of the year, but there has really been limited observation of just how individual fish use and compete for these limiting resources. Enter the study I am assisting with, where we seek to understand the subtleties behind how individuals seek out and defend cold water resources. At a basic level, our project involves observing brook trout behavior under a variety of thermal conditions. A population of brook trout, each tagged with a uniquely colored external tag, is placed into mock stream that we have created in the lab. The stream has three pools that we can manipulate to be different temperatures. Our goal is to compare individual interactions as stream temperature changes. So, I sit and watch, for endless hours, videos of fish being fish. I document how every individual interacts with every other individual in the stream and how much time fish spend in cold water vs. near food that is often in warm water. Why? Because we want to know how stable behavior is across temperatures. For example, we want to know how fish rank the importance of food vs. thermal refuge at various temperatures. We also want to see how fish interact among one another at different temperatures. We generally expect larger fish to rule the streams and dominate all other fish at cooler temperatures. But, when stream temperature heats up, larger fish become more stressed than smaller fish and may no longer be able to successfully compete for limiting resources. In short, big may not be better in warmer temperatures if you're a trout. While it may be a bit soon to comment on the data we have gathered so far, this project definitely holds promise to shed some new light on how individual behavior may shape the complex populations of brook trout that we know and love. We also hope to demonstrate just how important these limiting resources are for sustaining trout populations in thermally complex environments. Stay tuned!  I can't escape pipetting...not even while in the field. Here, I'm separating plasma off of red blood cells. I can't escape pipetting...not even while in the field. Here, I'm separating plasma off of red blood cells. It’s been awhile since I’ve posted a true research update. That’s because there’s not a lot going on. I mean, yes, I am working. And, I’m making good progress. But, not every day, or even week or month, leads to interesting results. But, I’m inching forward and I’ll reach the finish line eventually. Until then, I’m sparing you the details of how much pipetting and data entry I do on the average day. Trust me, it’s for your benefit. This week I was able to get out of the lab and into the field to do my monthly tissue collections. While it’s great to get into the field, this work is always a little anti-climactic. The objective of this study is to determine how fish respond to temperature stress at a cellular level. So, we go out and collect tissue samples, hoping to capture increases and decreases in gene expression, the measure of stress, as stream temperature rises and falls. Seems like I would have a carefully designed, clear sampling plan, right? Ha. My sampling dates are always a moving target. I try to predict stream temperature by looking at the five-day forecast. But, I’ve found that even obsessively checking all forecasting websites rarely gives a great prediction of air temperature or perception. Even if it did, air temperature doesn’t always predict stream temperature. But, it’s all I got. I then look at my calendar, try to scrounge up technicians and, voilà, I land on a date where it may or may not be the temperature I hoped for, but at least I have some help. Once I’m in the stream, I have no idea what the temperature actually is. I have loggers recording stream temperature every 30 minutes. But, I don’t download the data until the end of the day so that I know what temperature was while sampling. Doesn’t really matter, though. After a two-hour drive I would sample regardless of the stream temperature.  Just one of the trout footballs caught this week. The hatches are treating these fish very well. Just one of the trout footballs caught this week. The hatches are treating these fish very well. So, we collect 20 or so fish. Sometimes it’s an easy 4-5 hours, other times it’s a 10-hour fight battling high flows and small fish sizes. When I return back to campus, the samples go in a deep freeze and sit. Sometimes for a few days, sometimes for a few months. Once they are delivered to our collaborator in West Virginia who actually measures gene expression, the samples sit again. Most samples from 2016 have been process at this point, so I have some idea of what the data show. But, it’s still a guess, and I just cross my fingers we’re doing it right. So, I can’t tell you much about the data we are collecting. But, I can tell you, at least anecdotally, a little about how the populations are looking. Last summer was rough for trout. It was hot, it was dry, and our telemetry data showed that trout were getting picked out of the streams by birds and other animals left and right. There were then high rains in fall that washed away eggs, followed by a pretty mild winter. All in all, there was lot of concern in Pennsylvania about how the populations, particularly the young-of-years, were going to look this spring/summer. Turns out, they are doing just fine. At least in the handful of streams we are sampling right now. Unfortunately, the one-year-old fish are still a little too small for us to sample just yet, but they are huge. And, come November, these fish will be recruited into our study. More impressive, the young-of-year and little floating footballs. I’m not sure I’ve ever sampled a trout stream with fish that were consistently so large. (You also notice I’m not telling you where we’re sampling. It’s a public stream, but I’d like to keep all the fish to myself, thank you). Does that mean all the fish in Pennsylvania are doing well after the rough conditions over the last year? Absolutely not. The streams we sample are forested, have minimal fishing pressure, and at least one seems to have an exceptional forage base. In the weeks to come, a more extensive survey of Loyalsock streams will be undertaken by Susquehanna University. Until then, we won’t have a great picture on how Loyalsock populations faired over the last year. Stay tuned!  Home sweet, home for two summers. Camping for up to 14 days straight, this is where I learned to love field biology as an undergraduate. Home sweet, home for two summers. Camping for up to 14 days straight, this is where I learned to love field biology as an undergraduate. Ten years after my very first field season as an undergraduate, the field season that solidified my love for trout ecology, I have all the data I need for my Ph.D. dissertation. While I’m still working on a side project that occasionally allows me to get my feet wet, the time has finally come to hang up my waders and turn hard-earned data into results. This is the first summer in that time span that I’ve been home more than away. And, I don’t know how I feel about that. Sure, it’s great buying perishable groceries, taking hot showers, and having a roof over my head. And, I definitely don’t miss the unexplained bruises and deep soreness felt after carrying equipment that was a little too heavy for a little too long. But, I do miss the comradery of being in the field. For the last ten summers, I’ve put my friends and family aside and welcomed a whole new family into my life. My field family. People who often started as complete strangers, but who quickly learned my favorite foods, my quirky sleep habits, and every one of my pet peeves (including how to push them, when they wanted). Some joined the field family for only a few weeks as temporary technicians. Others were with me for several years and have since become more like members of my real family. From all I learned something, even if just bad habits and the drinking preferences of a retired army vet. There’s something special about a field family. Field partners see a completely different side of you that many of your closest friends and relatives may never know. I have a hard time describing why there is such a shift in demeanor, but the moment I begin packing the truck things change. It’s a mix of relaxation as you let go of any hopes of cell phone service and internet, but also a black cloud of anxiety as you’re trying to collect as much good data as possible despite constant equipment malfunctions and roadblocks. It doesn’t take long for pleasantries to fall to the wayside, particularly after you haven’t showered in days or seen another person outside of the crew. Everyone is always at some baseline level of exhaustion and frustration, but no one dare complain. Instead, you find laughter. You rib on each other, tell stories, and design elaborate practical jokes. At the end of the day, you squeeze out the last bit of sunshine doing various camp chores, preparing for the next day, and maybe even enjoying a few pages of a book. But, come nightfall, it’s just the crew and a campfire. And, let me tell you, campfire chat gets deep.  Typical field camp tent city as we gather around for the nightly fire. Typical field camp tent city as we gather around for the nightly fire. You spend a lot of time, celebrate a lot of holidays, and share a lot of experiences with your field crew. As a result, you’ll form a lot of memories with those people that only they will understand. A Corey Smith song playing while you’re driving around the backwoods of Virginia on a foggy evening. A YouTube video of movie outtakes from This Is 40. “Feesh, feesh.” Basement felonies. Fajitas. I can tell you the story behind every one of those, but it’s just not the same. Nostalgia aside, field partners are also people who you blindly trust to keep you safe. Entering into a field family is an unspoken pact that you will have the other person’s back at all times. And, you take it seriously. Great data can’t be collected when fearing for your safety. Unfortunately, one of the toughest lessons every biologist must learn is where the fine line is between “probably safe” and “that was really, really stupid.” While young scientists dance back and forth around the line often, I don’t think you ever truly learn to not cross it. So, the best you can do is trust that someone won’t put you into the stupid category too often, and will be able to safely get you out when they do. If you can find that level of trust, you’re guaranteed a great friendship. And, nothing will bring you closer than having to test it. Close calls are harrowing experiences, but make for great stories for many years to come.  Though he probably hated me here for going on this trip with a bum knee, Josh is the field partner, turned friend, turned family. Though he probably hated me here for going on this trip with a bum knee, Josh is the field partner, turned friend, turned family. Even when you’re not pushing the limits, accidents happen and the only way to recover is to come together. I tore my ACL walking next to a stream, and then refused surgery for over a year until my field seasons were complete. I could do that because I am insanely stubborn, but also because I knew my field partner would pick up my slack. He became the defacto person to hike the furthest, carry the most weight, and even support me when my knee starting popping out under the force from high velocity stream flows. Why? Because he’s a great person, but also because we shared a common goal. We needed the data. There’s something special about being part of a large crew that is willing to do whatever it takes to accomplish the goals and see each other succeed. So, yea, I’m excited that I can see a small light flickering at the end of the tunnel. But, you’ll have to excuse me if I’m feeling just a little sad to be entering into this next stage of my degree. The work may be mentally and physically exhausting, but many of my fondest memories and greatest achievements where made while in the stream with my field family. Our team is growing! At the end of the semester, Ben Kline, an undergraduate at Penn State, contacted the lab looking for volunteer opportunities. We're always happy to introduce prospective fish biologists to our field, but with limited field time I wouldn't blame any volunteer for losing interest in the work we're doing right now. Not only has Ben not lost interest, but he's taken it upon himself to collect some really great data looking at how individual fish compete for access to thermal refuge. Ben has volunteered to provide some guest posts over the summer, which will chronicle his experience in the lab and a little about what interests him in fisheries science. So, welcome Ben with his looks at Penns Creek, and learn more about him here.  A photo of the Kline family farm. A photo of the Kline family farm. Down along a stream called Penns Creek, there’s a place for me For as long as I can remember, Penns Creek has always been a part of my life. To be fair, I might be somewhat biased considering that my family farm has sat nestled on the bank of the Penns for over a hundred years. I often think back to my childhood sitting alongside the streambed, gazing into the murky water and just enjoying the quiet. If you have had the pleasure of spending some time on Penns Creek, then there is no denying that this location is certainly a lesser known gem of central Pennsylvania. Penns Creek is a tributary of the Susquehanna River, which ultimately empties into the Chesapeake Bay and has a long running history in the state of Pennsylvania. The original name “John Penn’s Creek” was named for William Penn’s younger brother, but was eventually adapted to simply “Penns Creek”. Penns Creek is considered to be Pennsylvania’s most impressive limestone stream in terms of size and length at a formidable 67.1-mile run. A limestone stream is characterized as being sourced primarily from groundwater, meaning that underground aquifers or springs are the source of water for the stream. This means the origin of the water feeding the stream could be countless miles from where the stream actually begins. Limestone streams are known to be shallow and slow moving, and typically are more resistant to changes in temperature once a certain set point is reached. The stream bottom consists primarily of gravel, mud, and sand substrate. Portions of the stream are littered with large boulders that add significant structure and habitat to the waterway and provide host to a variety of aquatic species.  The bridge that crosses Penns Creek to Little Mexico Campground from a kayak trip outside of New Berlin, Pa. The bridge that crosses Penns Creek to Little Mexico Campground from a kayak trip outside of New Berlin, Pa. The recreational use of Penns Creek is something that is well known to locals. Spending a day floating down the creek with your best buds or a nice day trip in the canoe or kayak sounds like the perfect way to spend a sunny Saturday in June. Aside from a nice paddle down the creek, Penns Creek also hosts countless opportunities for wildlife watchers with a large population of birds of prey, particularly Kingfishers and Bald Eagles. If birds aren’t for you, this is still a great place to catch a glimpse of painted turtles, muskrats, or even a mink along the streambank. While Penns Creek offers a great number of opportunities in recreation, perhaps the most noteworthy way to spend ones time on Penns Creek is fishing. Penns Creek is one of the most productive trout fisheries in all of Pennsylvania. However, the Penns is also host to a number of other desirable species such as smallmouth bass, various panfish, and even the occasional walleye. These legendary trout waters are known to host extremely high densities of trout in certain sections of the creek, of which some portions are even considered Class A Wild Trout streams. Anglers have even reported catches exceeding 20 inches in length in some of these locations.  My friend Logan trying to get in a few more casts in while fishing on Penns Creek as some kayakers pass by. My friend Logan trying to get in a few more casts in while fishing on Penns Creek as some kayakers pass by. There are a few contributing factors to this highly productive environment. The secluded nature of much of Penns Creek aids in the preservation of these natural populations, with some portions of the creek only accessible by foot, or in some cases, bicycle. These portions of the creek contain large rocks that make excellent trout habitat and help to form deep pools and riffles. The stands of old growth forest that hug the banks of Penns Creek provide an excellent source of refuge for hatching insects from heavy spring rains and predators, which allows for abundant food supply in these locations. The nature of a natural limestone stream is that which enables the water to support abundant natural life, which in this case, is a dense insect population. Droves of caddisfly, stonefly, and mayfly nymphs provide for these productive trout waters. Penns Creek is well known for its hatches, and perhaps the most famous of all is the green drake hatch, which is sought by anglers across the entire country. Habitat structure and food availability are two factors that make trout abundance high in Penns Creek; however, there is one resource that is becoming increasingly difficult to come by: cold water. Trout require thermal refuge, especially in the summer months when stream temperatures are known to rise significantly. In Penns Creek, the natural springs and seeping of cold groundwater provides cold habitat in the uppermost reaches of Penns Creek all the way into late August, which provides the trout with this limiting resource on a year-round basis. Despite the presence of consistent cold water in the upper reaches of the Penns Creek that make for excellent trout habitat, the middle and lower sections face significant thermal issues. As the water moves father from its source, the shallow and slow-moving nature of Penns Creek causes the water temperature to rise. As stated before, the nature of the stream makes Penns Creek highly resistant to temperature changes once a significant increase is reached. Since trout require cold water to thrive, many trout populations begin to seek cold water tributaries as refuge in these lower reaches of the Penns. This fragmented habitat may be the answer to sustaining these trout populations in times of thermal distress from rising stream temperatures. The lower third of the Penns becomes so warm in the summer that it is practically devoid of trout before it empties into the Susquehanna, and is now home to limited panfish and smallmouth bass populations only.  Natural springs like this help to cool warming stream water in the summer time. Natural springs like this help to cool warming stream water in the summer time. With climate change on the rise and cold water to support natural trout populations on the decline, we are currently seeking answers that may help us to better understand these growing concerns. I am currently working on a study that seeks to understand how individuals respond to a number of thermal conditions. This experiment hopes to identify what characteristics a fish might display that would make that individual more able to seek out and utilize these cold water refuges than others in a population. To do this, we are looking at a variety of data including movement, individual behavior, and genetics. The bulk of the work I am completing right now focuses on analyzing individual behavior and the interaction that individuals have with other members of the population. There is much more to share about this upcoming project, but I will save that for another post. A very exciting summer awaits, so stay tuned to find out about the gritty details of our new project in the weeks to come. |
AuthorShannon White Archives
October 2018
Categories
All
|
The Troutlook
A brook trout Blog
Proudly powered by Weebly




 RSS Feed
RSS Feed